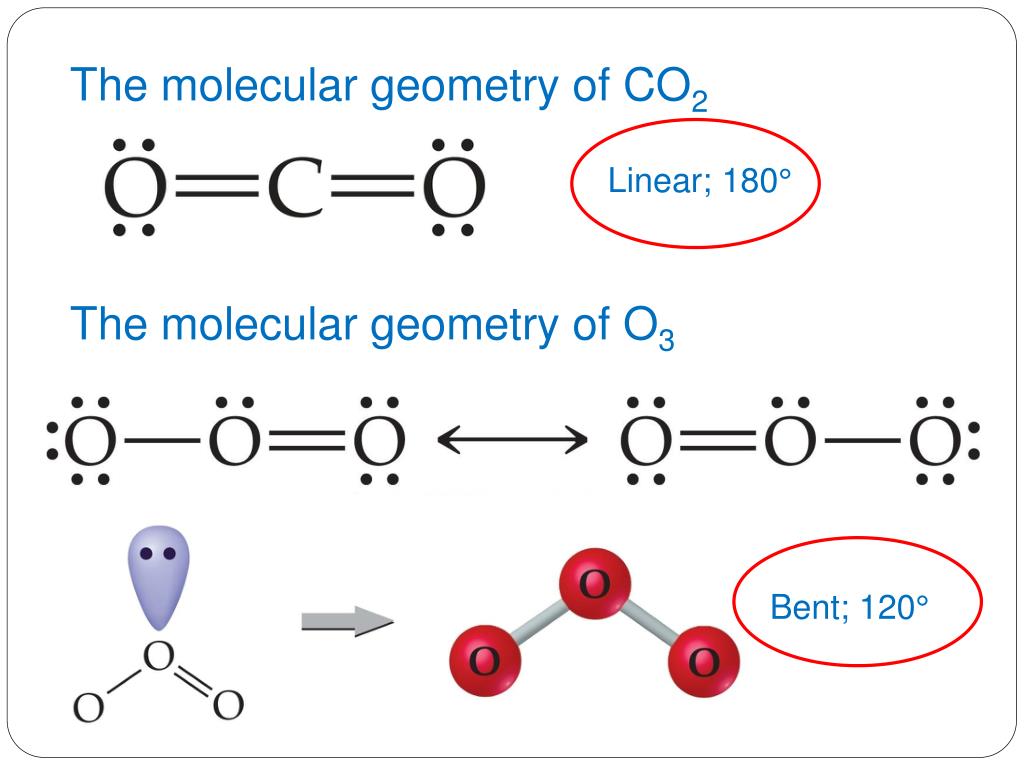
Two oxygen atoms are present at the terminals, where they share electrons and form bonds with the central carbon atom. Two double bonds connect the carbon and oxygen atoms in the Lewis structure. The carbon-oxygen ratio in a CO 2 molecule is 1:2. 
Since its creation, the Keeling Curve has served as a visual representation of Keeling’s data, which scientists have continued to collect since his death in 2005. Carbon dioxide is a colourless, odourless, incombustible gas produced by the combustion of carbon. By analyzing the CO2 in his samples, Keeling was able to attribute this rise to the use of fossil fuels. At Mauna Loa, he discovered global atmospheric CO2 levels were rising nearly every year. Keeling was able to establish a permanent residence at the Mauna Loa Observatory in Hawai'i, United States, to continue his research. He discovered CO2 levels are highest in the spring, when decomposing plant matter releases CO2 into the air, and are lowest in autumn when plants stop taking in CO2 for photosynthesis. By studying his measurements over the course of a few years, Keeling also noticed a larger seasonal pattern. Molecular Geometry (Shape) for CO2 (Carbon dioxide) Wayne Breslyn. the name of it is, arsenic pentachloride. this gives a complete orbit for As, but you need to place 6 dots on each chlorine to complete their orbit as well. He drew on his understanding of photosynthesis and plant respiration to explain this observation: Plants take in CO2 during the day to photosynthesize-or make food for themselves-but at night, they release CO2. What is the molecular shape and name AsCl5 the molecular shape is As is in the middle, where the 5 chlorine are surrounding it. We will use the D2h point group as a substitute since the orbital symmetries are retained in the D2h point group. The CO 2 molecule is linear and its point group is Dh. 1: Bond distances (lengths) and angles are shown for the formaldehyde molecule, H2CO. Bond distances are measured in Ångstroms (1 Å 10 10 m) or picometers (1 pm 10 12 m, 100 pm 1 Å). H 2 O Water NH 3 Ammonia CH 4 Methane SF 6 Sulfur Hexafluoride SF 4 Sulfur Tetrafluoride PF 5 Phosphorus Pentafluoride ClF 3 Chlorine Trifluoride Polymers A Level. In sulphur dioxide, as well as the two double bonds, there is also a lone pair on the sulphur. In the carbon dioxide, the two double bonds try to get as far apart as possible, and so the molecule is linear. Carbon dioxide is linear, while sulphur dioxide is bent (V-shaped). 
The air samples taken at night contained a higher concentration of CO2 compared to samples taken during the day. Find the point group of the molecule and assign Cartesian coordinates so that z is the principal axis. A bond distance (or bond length) is the distance between the nuclei of two bonded atoms along the straight line joining the nuclei. SHAPES OF MOLECULES AND IONS (including double bonds) 1. Keeling began studying atmospheric carbon dioxide in 1956 by taking air samples and measuring the amount of CO2 they contained. The Keeling Curve is named after its creator, Dr. The Keeling Curve is a graph that represents the concentration of carbon dioxide (CO2) in Earth’s atmosphere since 1958.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
